 
The net sum of valid resonance structures is defined as a resonance hybrid, which represents the overall delocalization of electrons within the molecule. Resonance structures are used when one Lewis structure for a single molecule cannot fully describe the bonding that takes place between neighboring atoms relative to the empirical data for the actual bond lengths between those atoms. It compares and contrasts two or more possible Lewis structures that can represent a particular molecule. Resonance is a mental exercise and method within the Valence Bond Theory of bonding that describes the delocalization of electrons within molecules. Structural formulas for molecules involving H, C, N, O, F, S, P, Si, ClĮlements in the same column (group) of the periodic table are similar to one another resulting in similar structural formulas:ĭelocalized Electrons and Resonance in Ions and Molecules All the atoms are geometrically equivalent with bond angles of 90°. 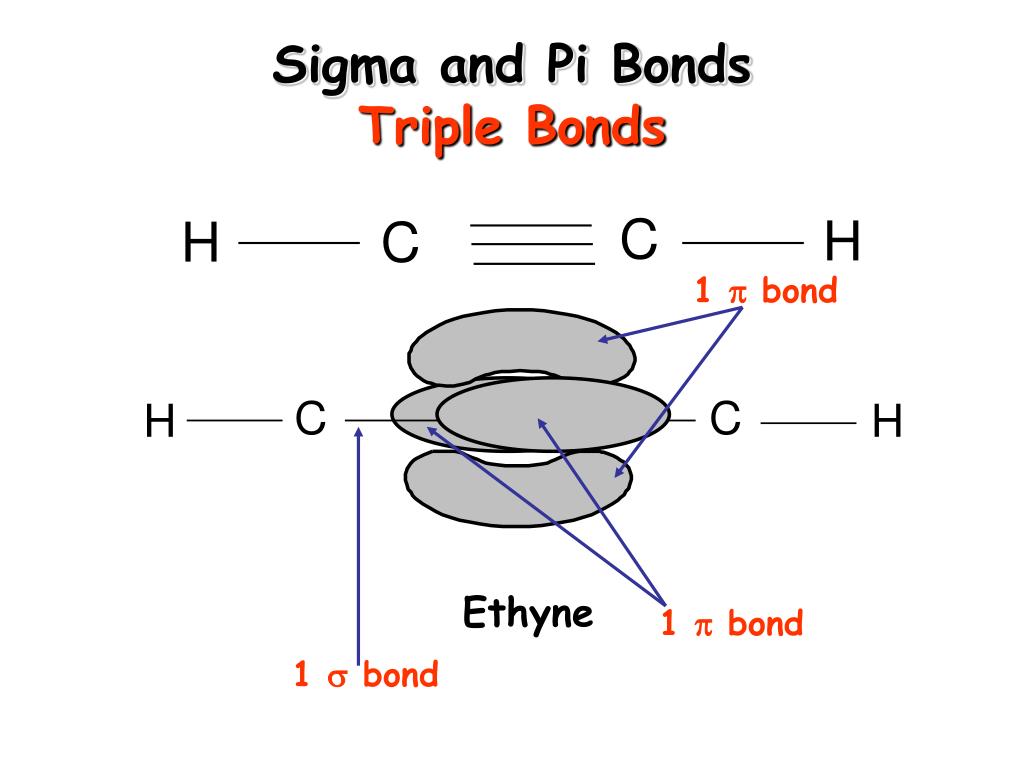 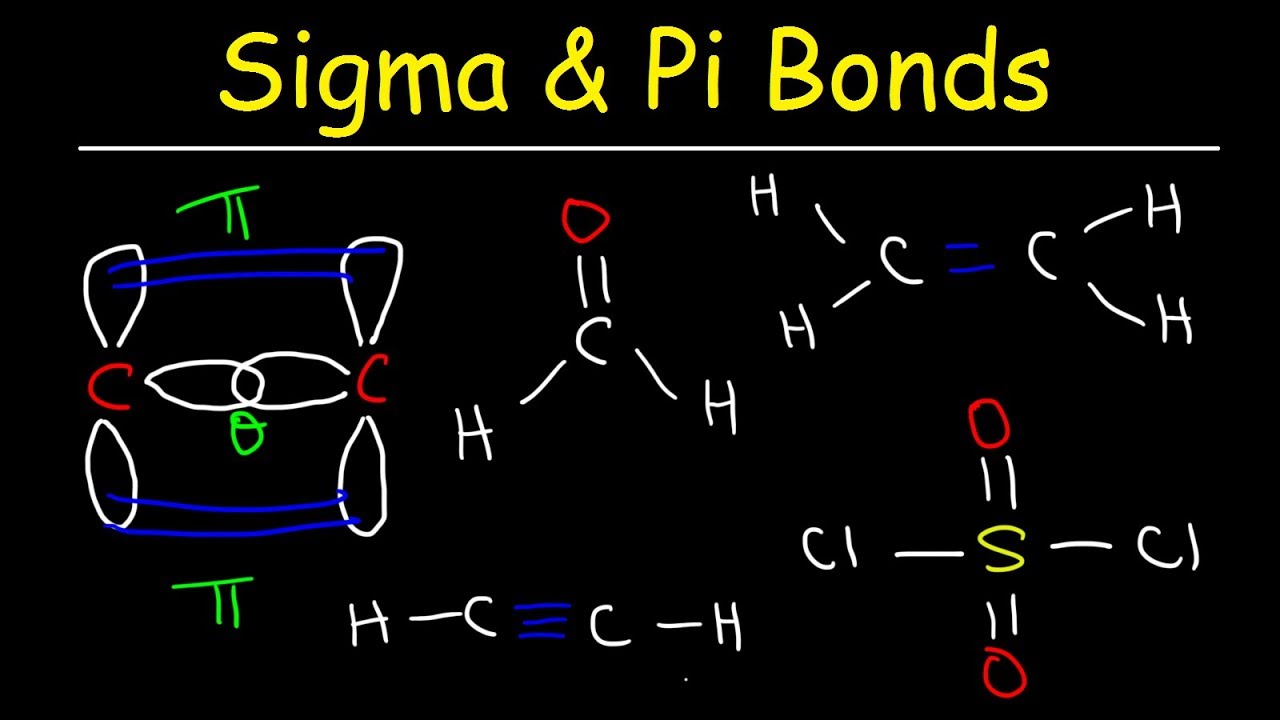
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
